US FDA accepts first biologics license application for AstraZeneca’s durvalumab in bladder cancer
FDA grants priority review status with PDUFA set for Q2 2017

FDA grants priority review status with PDUFA set for Q2 2017

Den första randomiserade fas 3-studien (AURA3) som jämför osimertinib med dubbel platinumbaserad cytostatikabehandling Osimertinib förbättrade progressionsfri överlevnad med nästan sex månader Patienter med metastaser på centrala nervsystemet visade på liknande resultat som den totala populationen enligt förbestämda subgruppsanalyser

Tagrisso scientific updates include results of AURA3 trial in patients with lung cancer, and Phase II efficacy data on CNS metastases New analyses of Phase III FALCON trial will extend understanding of Faslodex in 1st-line treatment of advanced breast cancer New acalabrutinib safety and efficacy data in patients with difficult-to-treat chronic lymphocytic leukaemia

AstraZeneca today announced that the US FDA has lifted the partial clinical hold on the enrolment of new patients with head and neck squamous cell carcinoma (HNSCC) for clinical trials of durvalumab as monotherapy and in combination with tremelimumab or other potential medicines.

Korta fakta om diabetes - en växande folksjukdom. AstraZeneca har som mål att förbättra livet för personer med diabetes genom att minska påverkan av sjukdomen och dess komplikationer. Vi forskar inom metabola sjukdomar och har en ambition att vara ledande på området. AstraZeneca erbjuder olika behandlingsmöjligheter inom typ 2-diabetes.

Resultatet för tredje kvartalet är i linje med våra förväntningar

Trial studied Lynparza as maintenance treatment for women with BRCA-mutated metastatic ovarian cancer Initial findings show safety profile with Lynparza tablets was consistent with previous studies

AstraZeneca today announced that the US Food and Drug Administration (FDA) has accepted a complete re-submission of a New Drug Application (NDA) for sodium zirconium cyclosilicate (ZS-9), a potential new medicine for the treatment of hyperkalaemia (high potassium level in the blood serum) by ZS Pharma, a wholly-owned subsidiary of AstraZeneca. The FDA has indicated that this is a complete class 2
AstraZeneca today announced that it has entered into an agreement with Cilag GmbH International, an affiliate of Johnson & Johnson, for the divestment of the rights to Rhinocort Aqua outside the US.
AstraZeneca today announced that it has entered into an agreement with Aralez Pharmaceuticals Trading DAC, a subsidiary of Aralez Pharmaceuticals Inc., for the rights to branded and authorised generic Toprol-XL (metoprolol succinate) in the US
Brilinta did not demonstrate a benefit over clopidogrel in a symptomatic peripheral artery disease patient population
AstraZeneca today announced that MedImmune, its global biologics research and development arm, has entered into a licensing agreement with Allergan plc for the global rights to MEDI2070.
Phase III FALCON trial demonstrates superiority of Faslodex over Arimidex in the 1st-line treatment of hormone-receptor positive (HR+) advanced breast cancer Immuno-Oncology data reinforce commitment to lung and head and neck cancers
AstraZeneca today announced its decision to withdraw the Marketing Authorisation Application (MAA) submitted to the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) in June 2015 for cediranib in combination with platinum-based chemotherapy
DURATION-8 trial met primary and secondary endpoints, significantly reducing blood sugar (HbA1c), weight and systolic blood pressure versus either medicine alone The first trial to assess GLP-1RA/SGLT-2i combination therapy
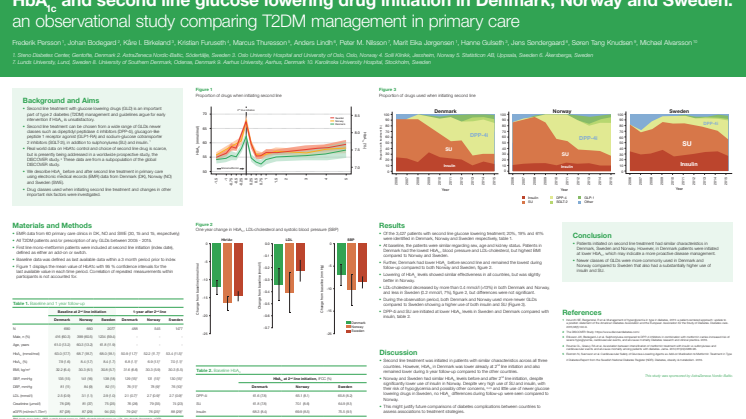
Kontrollen av blodsocker och kolesterol hos patienter med typ 2-diabetes är bättre i Danmark jämfört med i Norge och i Sverige enligt resultat från studien Nordic DISCOVER. Studien är den första nordiska jämförande långsiktiga studien av primärvårdens omhändertagande av patienter med typ 2-diabetes. Resultat presenteras för första gången på den europeiska diabeteskonferensen EASD, 14 september.
Sjukdomskostnaderna för behandling av patienter med typ 2-diabetes fördubblades i Sverige mellan åren 2006 och 2014 samtidigt som kostnaderna för diabetesläkemedel genomgående under perioden endast representerar fyra procent. Det visar resultat från den svenska studien DAISY som presenteras på den europeiska diabeteskonferensen EASD, den 13 september.
First programme to assess potential cardiovascular and renal benefit of an SGLT-2i beyond diabetes
AstraZeneca’s first biologic respiratory medicine met primary and key secondary endpoints in pivotal trials for severe asthma
New evidence to be presented includes pivotal Phase III data on benralizumab