
Scandinavian Biopharma welcomes Sara Grahm to the position as Director of Manufacturing
Scandinavian Biopharma continues to strengthen its organization in manufacturing and is pleased to welcome Sara Grahm as our new Director of Manufacturing.

Scandinavian Biopharma continues to strengthen its organization in manufacturing and is pleased to welcome Sara Grahm as our new Director of Manufacturing.

Scandinavian Biopharma continues to grow and we also start this year by welcoming new colleagues. We are further strengthening in manufacturing and are pleased to welcome Maria Edlund as our Drug Product Specialist.

For several years we have supported the orphanages in Grand Popo by donating food, medicine and other supplies to improve the situation of the children. Our Seasonal gift will guarantee that the children at the orphanages have a proper supply of rice, beans, spaghetti, tomatoes and cooking oil. We are committed to support children in low- and middle-income countries.

We are expanding the business so much that we need larger premises. During the pandemic, the company has hired many new employees in research, clinical development, registration, manufacturing and sales. The space is specially adapted for Scandinavian Biopharma's operations and has a tailor-made, state of the art laboratory with a process development unit as well as a modern meeting place.

Scandinavian Biopharma has strengthened its vaccine portfolio by the addition of an innovative preclinical project for the development of a new Campylobacter jejuni vaccine. Scandinavian Biopharma will have the global rights for a Campylobacter jejuni vaccine technology developed by VaxAlta.

An improved user-friendly formulation of the world's most advanced ETEC vaccine candidate, ETVAX®, has been developed to meet commercial requirements. We want to ensure that the new formulation gives rise to immune responses that are non-inferior (i.e. not worse) to the previous formulation of the vaccine that underwent earlier testing. This is done in a phase II double-blinded, randomized study.

Scandinavian Biopharma continues to grow and welcome new colleagues. We are now further strengthening our organisation in manufacturing and are pleased to welcome Aino Råberg as our Drug Substance Lead.

The Vaccines Against Shigella and ETEC (VASE) Conference, on September 28 & 30, will provide key updates on vaccines being developed for Shigella, ETEC, and other emerging diarrheal pathogens. 2 of the 18 presentations at VASE is highlighting our ETEC vaccine candidate ETVAX®.

Scandinavian Biopharma continues to expand and strengthen its quality organization. Camilla Jidling has been hired as the new Quality Assurance Manager and has been working at Scandinavian Biopharma since the beginning of the summer.

Welcome Fernanda, to our team at Scandinavian Biopharma.

We are pleased to welcome Günes Uysal to our team at Scandinavian Biopharma.
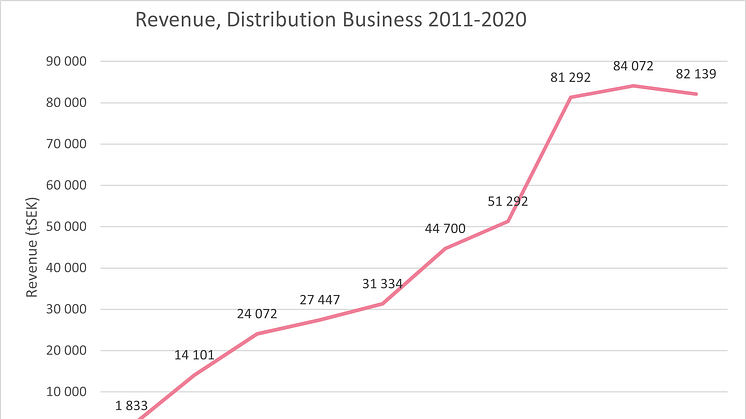
The year we have left behind us has been different in many ways. Despite the major impact the pandemic had on operations in 2020, Scandinavian Biopharma reports largely unchanged revenue for the distribution operations and important progress has been made in the development of the ETEC vaccine candidate ETVAX®.
Vi utvecklar det första vaccinet i världen mot diarré orsakad av ETEC för att skydda både barn och vuxna i endemiska länder och resenärer till högriskdestinationer.
Vi distribuerar ett brett sortiment av biologiska specialistläkemedel med fokus på vacciner och immunglobuliner.
www.scandinavianbiopharma.se
Vår information på mynewsdesk riktar sig till journalister och media.
