Press release -
Mechanism Behind Onset of Autoimmune Pancreatitis Caused by Changes in Intestinal Flora Elucidated Development of New Treatment Methods Expected - Kindai University
1. Highlights of Study
- Elucidation of the influence of intestinal flora on mechanism behind the onset of autoimmune pancreatitis
- Elucidation of the mechanism behind the changing of intestinal flora controlling the onset of autoimmune pancreatitis
- Expected to lead to developments of new treatment methods of autoimmune pancreatitis
2. Content of the Research
Chronic inflammation of the pancreas is broadly classified into two diseases; autoimmune pancreatitis and chronic pancreatitis. Autoimmune pancreatitis, an intractable disease, causes the autoimmune system to mistakenly attack the pancreas causing systemic inflammation. Also, it can cause secondary diabetes and all sorts of cancer including pancreatic.
The disease is said to be common amongst elderly men, and the particulars of the mechanism of its onset were not clear. Currently, steroids are used in immunosuppressive therapy for autoimmune pancreatitis, but steroids have various side effects, so the development of new treatments has been hoped for. Prof. Watanabe’s team has been continuously conducting research on autoimmune pancreatitis and chronic pancreatitis since 2008.
In this study on autoimmune pancreatitis, focus was placed on changes in the intestinal immune system that accompanied changes in intestinal flora. Experiments were conducted on how these changes affected the onset of autoimmune pancreatitis of the pancreas. By artificially changing the intestinal flora of mice, we found that the number of plasmacytoid dendritic cells*1, a type of immune cell, increased in the pancreas.
This result suggests that the development of autoimmune pancreatitis is largely related to the immune response to intestinal bacteria. The results of this research not only revealed that the immune response to intestinal bacteria is involved in the mechanism of the onset of autoimmune pancreatitis, but it is also expected to lead to the development of new treatment and prevention methods.
https://www.cell.com/trends/immunology/fulltext/S1471-4906(18)30170-4
This paper further adds to developments in research of, and introduces the results of research specifically on autoimmune pancreatitis.
3. About Publication
Research paper title: Intestinal dysbiosis mediates experimental autoimmune pancreatitis via activation of plasmacytoid dendritic cells
Journal name: International Immunology (IF:5.189)
Co-authors: Ken Kamata, Tomohiro Watanabe*, Kousuke Minaga, Akane Hara, Tomoe Yoshikawa, Ayana Okamoto, Kentaro Yamao, Mamoru Takenaka,
Masatoshi Kudo, Clinical Internal Medicine (Gastroenterology) at the Faculty of Medicine of Kindai University
Ah-Mee Park, Microbiology at the Faculty of Medicine of Kindai University
*Corresponding author
Prof. Watanabe’s research group have been working on the elucidation of the onset mechanism of autoimmune pancreatitis and chronic pancreatitis since 2008; a review paper of research results has been published in ‘Trends in Immunology’.
4. Details of Research
About 100 trillion bacteria, called intestinal flora, are said to be present in the human intestinal tract maintaining a certain balance. In recent years, discoveries have shown that the intestinal flora control the immune response in the intestinal tract, and is related not only to those in the intestinal tract, but in the onset of various systemic diseases. Watanabe’s research group have reported that plasmacytoid dendritic cells producing type I interferon*2 and interleukin 33*3 play a pathological role in the onset of autoimmune pancreatitis. (Trends in Immunology 2018; 39: 874-889, Journal of Immunology 2017; 198: 3868-3896, Journal of Immunology 2015; 195: 3033-3044). However, to the question, “What kind of stimulation causes plasmacytoid dendritic cells to produce type I interferon and interleukin 33?” until now the mechanism has not been made clear.
Therefore, the research group hypothesized that “a change in intestinal flora could activate plasmacytoid dendritic cells and cause disease” and proceeded with the research. In the first experiment the intestinal bacteria of autoimmune pancreatitis model mice were eliminated by administering antibiotics - the intestinal flora was dramatically changed - the number of plasmacytoid dendritic cells present in the pancreas decreased, and the onset of autoimmune pancreatitis being clearly suppressed was observed. Also, using a next generation sequencer to examine the intestinal flora of the mice revealed that the mice that had developed autoimmune pancreatitis had lost intestinal flora diversity compared to normal mice. These findings indicate that changes in intestinal flora affect the onset of autoimmune pancreatitis.
Over the course of further research, it was established that translocation of intestinal flora in mice that developed autoimmune pancreatitis, developed autoimmune pancreatitis even under conditions the disease would not develop normally. Further, it was discovered that transplantation of feces from mice with autoimmune pancreatitis promoted development of autoimmune pancreatitis. Of great interest, with changes in the intestinal flora, there was a large increase of plasmacytoid dendritic cells producing type I interferon and interleukin 33 in the pancreas. From the above results, it is clear that a change in intestinal flora is one factor that promotes the onset of autoimmune pancreatitis.
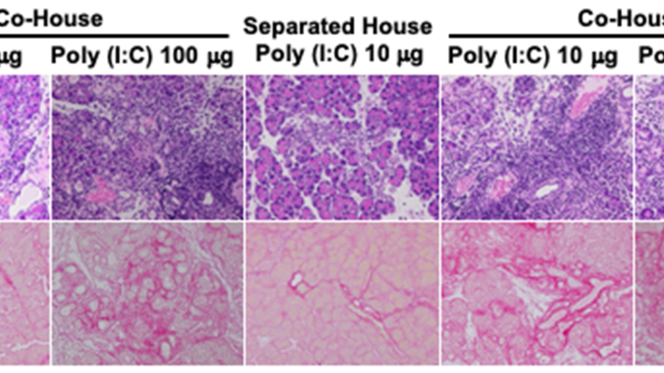
Top row: Pancreatic inflammation observed by hematoxylin & eosin staining
Bottom row: pancreatic fibrosis observed by sirius red staining
Detailed description of diagram:
Autoimmune pancreatitis can be induced in MRL/ MpJ mice by repeatedly administering Poly (I:C) (a synthetic double-stranded RNA, which has an equivalent immunostimulatory effect as virus-derived double-stranded RNA). Although the onset of autoimmune pancreatitis usually requires administration of 100μg of Poly (I:C), when the mice are put under conditions where intestinal bacteria is translocated, only 1/10th the amount of Poly (I:C) needs to be administered (10μg), causing intense autoimmune pancreatitis. Separated house; reared under conditions that do not cause translocation of intestinal bacteria. Co-House; reared under conditions that cause translocation of intestinal bacteria.
5. Glossary
1. Plasmacytoid dendritic cell
An immune cell. Dendritic cell that specializes in producing type I interferon.
2. Type I interferon
A type of cytokine (protein) secreted by cells. It is frequently produced in cases of microbial infection, especially in the case of viral infection. It plays an important role in immune defense during microbial infection. In contrast, in the case of no microbial infection, excess production leads to onset of autoimmune disease.
3. Interleukin 33
A type of cytokine (protein) secreted by cells. It plays an important role in inflammations, allergies, and fibrosis (abnormal tissue growth).
6. Future Outlook
This research unveiled the strong relationship between changes of the intestinal flora and onset of autoimmune disease. It is expected that use of “intestinal flora / type 1 interferon / Interleukin 33” as controls will lead to development of new treatment methods of auto-immune pancreatitis.
